 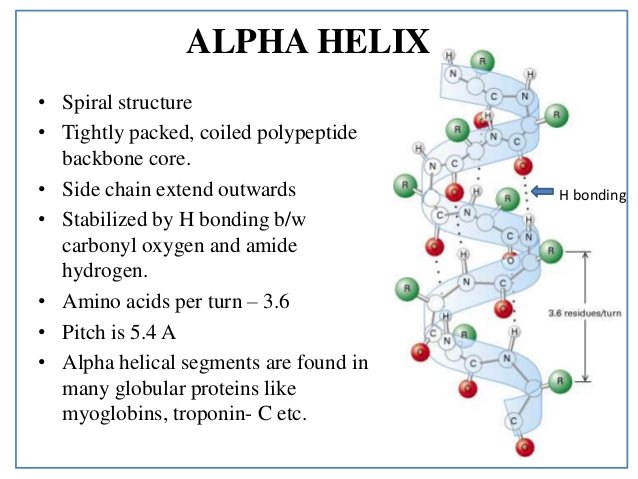
What's wrong with the diagram? Two things:įirst of all, only the atoms on the parts of the coils facing you are shown. We'll talk it through in some detail after you have had a look at it. This is a very simplified diagram, missing out lots of atoms. 
The next diagram shows how the alpha-helix is held together by hydrogen bonds. The "alpha" means that if you look down the length of the spring, the coiling is happening in a clockwise direction as it goes away from you. In an alpha-helix, the protein chain is coiled like a loosely-coiled spring. This is obviously a potential source of confusion! This is another covalent link and so some people count it as a part of the primary structure of the protein.īecause of the way sulphur bridges affect the way the protein folds, other people count this as a part of the tertiary structure (see below). If two cysteine side chains end up next to each other because of folding in the peptide chain, they can react to form a sulfur bridge. Obviously, all the peptide links are made of covalent bonds, so that isn't a problem.īut there is an additional feature in proteins which is also covalently bound. The wider definition of primary structure includes all the features of a protein which are a result of covalent bonds. The N-terminal is always written on the left of a diagram for a protein's primary structure - whether you draw it in full or use these abbreviations. If you followed the protein chain all the way to its left-hand end, you would find an amino acid residue with an unattached -NH 2 group. If you look carefully, you will spot the abbreviations for glycine (Gly) and alanine (Ala) amongst the others. Using three letter abbreviations, a bit of a protein chain might be represented by, for example: These abbreviations commonly consist of three letters or one letter. This primary structure is usually shown using abbreviations for the amino acid residues. In other words, if you replaced the "R" groups in the last diagram by real groups you would have the primary structure of a particular protein. At its simplest, the term is used to describe the order of the amino acids joined together to make the protein. Now there's a problem! The term "primary structure" is used in two different ways. The peptide chain is known as the backbone, and the "R" groups are known as side chains. The "R" groups come from the 20 amino acids which occur in proteins. The end of the peptide chain with the -NH 2 group is known as the N-terminal, and the end with the -COOH group is the C-terminal.Ī protein chain (with the N-terminal on the left) will therefore look like this: The unchanged -COOH group is written at the right-hand end. The peptide chain is made up from what is left after the water is lost - in other words, is made up of amino acid residues.īy convention, when you are drawing peptide chains, the -NH 2 group which hasn't been converted into a peptide link is written at the left-hand end. When the amino acids combine together, a water molecule is lost. You have to use this term because strictly speaking a peptide chain isn't made up of amino acids. If you joined lots and lots together (as in a protein chain), you get a polypeptide.Ī protein chain will have somewhere in the range of 50 to 2000 amino acid residues. If you joined three amino acids together, you would get a tripeptide. In chemistry, this would also be known as an amide link, but since we are now in the realms of biochemistry and biology, we'll use their terms. In each case, the linkage shown in blue in the structure of the dipeptide is known as a peptide link. It is possible for this to happen in one of two different ways - so you might get two different dipeptides. Glycine and alanine can combine together with the elimination of a molecule of water to produce a dipeptide. That means that the two simplest amino acids, glycine and alanine, would be shown as: It is much easier to see what is happening if you do that. However, for drawing the structures of proteins, we usually twist it so that the "R" group sticks out at the side. 
In chemistry, if you were to draw the structure of a general 2-amino acid, you would probably draw it like this: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
